AI Startups
The $45 Million Per Month Bottleneck: How Ritivel AI Automates Drug Submissions


TL;DR
Discover how Ritivel AI is solving the pharmaceutical industry's multi-billion dollar regulatory bottleneck with highly traceable generative AI tailored for life sciences.
The Billion-Dollar Bottleneck in Drug Development
Life sciences runs at two different speeds.
On the research side, in silico modeling and computational biology have genuinely changed the pace, drug targets get identified faster, assays that used to need wet lab time can be simulated and discovery timelines that once runs faster.
Regulatory submissions haven't. Teams of experienced writers still manually convert thousands of pages of trial data into precisely formatted documents that satisfy global standards like the Common Technical Document.
Clinical Study Reports take 8 to 12 weeks each. Every month a drug spends clearing that process costs a pharma company roughly $45 million in lost revenue. Patients wait longer for treatments that have already demonstrated they work.
Why Generic LLMs Don't Fix This
AI is the obvious solution. LLMs write fluently, summarize dense documents, can imitate regulatory tone. So why haven't they made a dent in submissions?
The FDA and EMA require every clinical claim in a submission to trace back to a specific source: a trial protocol, a statistical output, a reference document. Standard models can't maintain that chain. They generate plausible text from patterns, and when they're wrong, there's no signal. A hallucinated study citation, a confidence interval off by a decimal, an inclusion criterion slightly garbled—none of it throws an error. In a submission, any of those can mean a full rework. Regulatory teams live with enough deadline pressure without adding a new category of undetectable error.
The problem isn't AI writing quality. It's a specific technical gap that requires no traceability.
What Ritivel Built
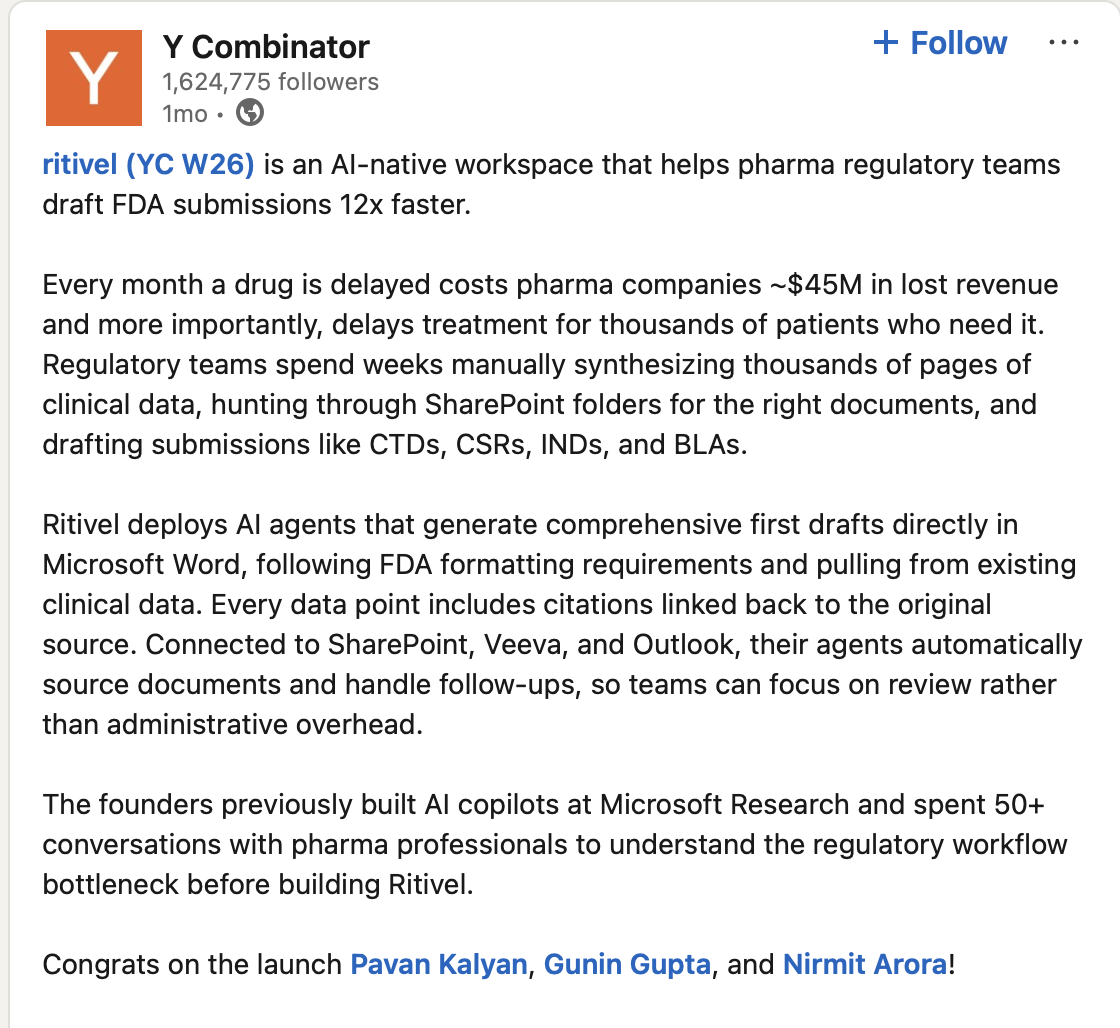
Ritivel AI—founded in 2025, Y Combinator-backed—came out of Microsoft Research's AI copilot work. Before building anything, they spent months talking to pharma regulatory, biostatistics, and clinical ops teams. The consistent problem wasn't "we need better writing tools." It was more specific: taking structured clinical trial data and turning it into submission-grade documents, fast, with every claim auditable.
They built for that exactly. An AI workspace that drafts CTDs, CSRs, INDs, and BLAs. Writers review and refine instead of producing from scratch.
Four Things That Make It Actually Usable
To get past a pharma company's procurement and legal teams, an AI tool has to offer more than just speed. Ritivel relies on four strong pillars:
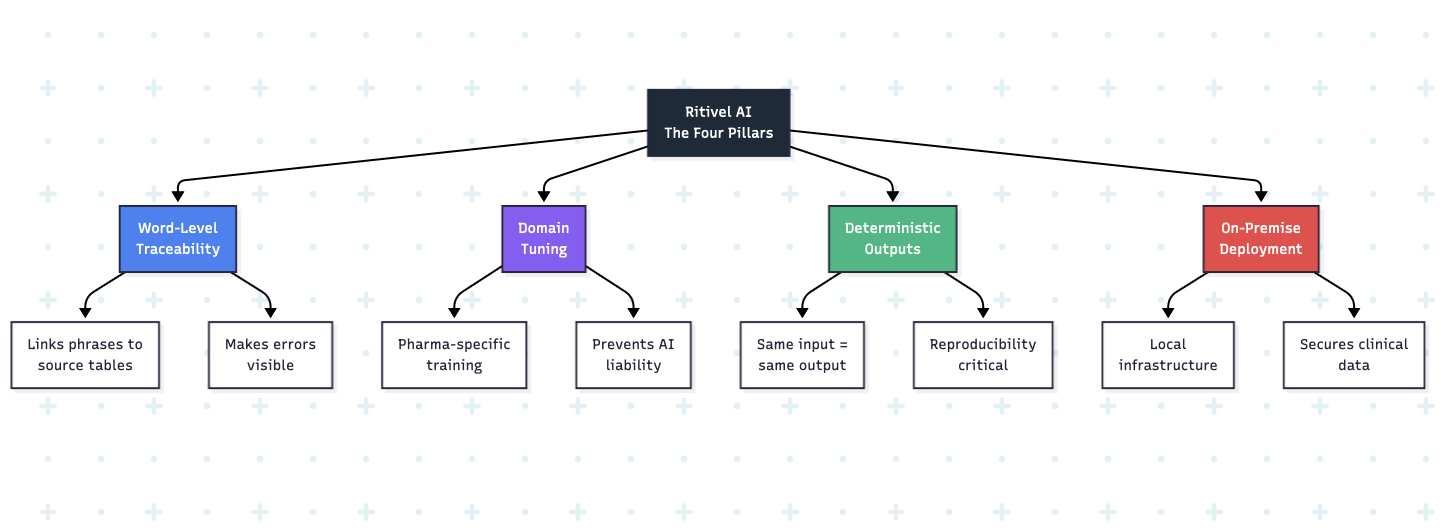
Word-level traceability: Every generated paragraph links specific phrases directly back to source tables, prior submissions or regulatory guidance. Reviewers can instantly click to verify whether a conclusion is genuinely supported by the underlying data. Errors become immediately visible instead of buried in the text.
Deterministic outputs: Same dataset = Same output. Documents submitted today might face regulatory scrutiny years down the line, meaning reproducibility matters infinitely more than creative variation.
Domain tuning: Models trained on broad internet data don't understand the rigorous evidentiary standards of pharmaceutical writing. In this industry, a generic model is a massive liability.
On-premise deployment: Most AI tools operate as multi-tenant cloud SaaS. For pharma, sending proprietary clinical data and draft submissions into the cloud is usually a hard no for IT security. Ritivel runs entirely inside the customer's own infrastructure, completely removing a major adoption blocker.
The Practical Picture
Regulatory automation is still a relatively thin field. Pharma companies staring down the barrel of patent cliffs and tightening global regulations simply don't have the patience for submissions that get delayed by weeks just because a writer is manually reformatting data tables into prose.
The tools that can actually compress that timeline without adding compliance exposure will inevitably get bought and deeply embedded into enterprise workflows. The ones that introduce compliance risks will get killed at the legal review stage, regardless of how impressive their initial demo looked.
Thankfully, Ritivel looks promising.